The Genetic Architecture of Behavioral Disorders
Exploring the Role of Epigenetics in Psychiatry
Kirolos Eskandar
Psychotherapie-Wissenschaft 15 (1) 2025 57–63
www.psychotherapie-wissenschaft.info
https://doi.org/10.30820/1664-9583-2025-1-57
Abstract: Behavioral disorders such as depression, schizophrenia, ADHD, and autism spectrum disorders represent a significant challenge in psychiatry due to their complex and multifactorial nature. While traditional models have focused on genetic predispositions, emerging research highlights the critical role of epigenetics in modulating gene expression in response to environmental factors. This literature review explores the genetic architecture of behavioral disorders, emphasizing the influence of epigenetic mechanisms such as DNA methylation, histone modification, and non-coding RNAs. We delve into how these processes contribute to the onset, progression, and transgenerational transmission of psychiatric conditions. Furthermore, we discuss the implications of these findings for personalized treatment strategies and future research directions, underscoring the potential for epigenetic therapies in reshaping psychiatric practice. This review aims to bridge the gap between genetics and environment, offering a comprehensive understanding of how epigenetics can inform the future of psychiatric care.
Keywords: behavioral disorders, epigenetics, genetic architecture, psychiatry, personalized medicine
Behavioral disorders, such as ADHD, autism spectrum disorder (ASD), schizophrenia, and mood disorders, are common in psychiatric practice and have traditionally been understood through a combination of clinical observation, psychological theory, and, more recently, neurobiology. These disorders are often characterized by symptoms such as inattentiveness, hyperactivity, social communication difficulties, delusions, hallucinations, and mood dysregulation, which can vary widely in severity and impact on daily functioning.
The traditional understanding of these disorders has largely been rooted in diagnostic frameworks such as the DSM-5, which categorizes them based on observed behaviors and reported symptoms. For example, ADHD is defined by persistent patterns of inattention and hyperactivity, while schizophrenia is characterized by psychotic symptoms such as delusions and hallucinations. Historically, psychiatric models have focused on the role of neurotransmitter imbalances, brain structure abnormalities, and environmental factors in the development and manifestation of these disorders.
However, recent advancements in genetics and molecular biology have prompted a paradigm shift in the understanding of these conditions. Research has increasingly highlighted the complex genetic architecture underlying behavioral disorders, pointing to the role of multiple genes and their interactions with environmental factors in the etiology of these conditions. For instance, studies have shown that ADHD, ASD, and schizophrenia share several genetic risk factors, suggesting that these disorders may lie on a spectrum of neurodevelopmental conditions with overlapping biological pathways (Andreassen et al., 2023; Guo et al., 2024).
Moreover, the emerging field of epigenetics—whereby gene expression is altered without changes to the underlying DNA sequence—has introduced new perspectives on how environmental factors can influence genetic predispositions. Epigenetic modifications, such as DNA methylation and histone modification, can be triggered by various environmental factors, including stress, diet, and exposure to toxins, potentially leading to the development of psychiatric disorders in genetically predisposed individuals. This has led to a growing recognition of the need to integrate genetic and epigenetic insights into psychiatric models to better understand, diagnose, and treat these complex disorders (Pina-Camacho et al., 2016; Ribasés et al., 2023).
A systematic approach was utilized for this literature review, adhering to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines to gather relevant articles and studies in Emergency medicine’s critical cases. A thorough search was conducted in reputable databases, including PubMed, Google Scholar, Scopus, and Web of Science, using specific keywords such as «Behavioral Disorders», «Epigenetics», «Genetic Architecture», «Psychiatry», and «Personalized Medicine» to ensure comprehensive coverage of pertinent literature.
The inclusion criteria for the studies were as follows: (1) publications in English, (2) studies focusing specifically on epigenetics in psychiatry, and (3) studies reporting on genetic architecture of behavioral disorders. Initially, 127 articles were retrieved from the databases. After a meticulous examination to eliminate duplicate references, 41 unique articles met the inclusion criteria. These articles underwent rigorous evaluation through a comprehensive assessment of their titles, abstracts, and full texts, confirming their alignment with the established inclusion criteria and warranting their inclusion in the review.
To provide a clear overview of the study selection process, the PRISMA flow diagram is included below (fig. 1), illustrating the number of records identified, screened, and included in the review, along with reasons for exclusion at each stage.
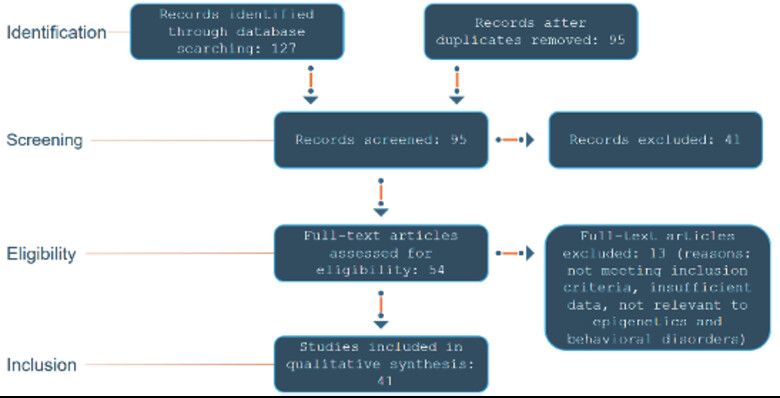
Fig. 1: PRIMSA flow diagram
The Genetic Basis of Behavioral Disorders
The genetic basis of behavioral disorders is increasingly understood through the lens of genomic research, which has elucidated the role of genetic variations in the development and expression of psychiatric conditions. Behavioral disorders such as ADHD, autism spectrum disorder (ASD), schizophrenia, and mood disorders have long been recognized as heritable, with family and twin studies consistently demonstrating significant genetic components. However, the precise genetic underpinnings have only become clearer with advances in genomic technology, particularly through the use of genome-wide association studies (GWAS).
GWAS have identified numerous genetic loci associated with behavioral disorders, highlighting the polygenic nature of these conditions. For instance, studies have found that variations in the DRD4 and DAT1 genes, which are involved in dopamine regulation, are linked to ADHD, a disorder characterized by inattention and hyperactivity (Brainstorm Consortium et al., 2018). Similarly, mutations in the SHANK3 gene, which plays a critical role in synapse formation, have been implicated in some cases of ASD, a disorder marked by challenges in social communication and repetitive behaviors (Wang et al., 1998).
Schizophrenia, another complex psychiatric condition, has been associated with the COMT gene, which influences the degradation of dopamine in the brain, and the DISC1 gene, involved in brain development (Pettersson et al., 2016). These genetic findings underscore the complexity of behavioral disorders, which often result from the interaction of multiple genes rather than single-gene mutations.
GWAS have been particularly instrumental in revealing the shared genetic architecture between different psychiatric disorders. For example, studies have shown that schizophrenia and bipolar disorder share common genetic risk factors, suggesting a biological overlap between these two conditions (Brainstorm Consortium et al., 2018). Additionally, GWAS have identified significant loci on chromosome 6, specifically within the Major Histocompatibility Complex (MHC) region, that are associated with schizophrenia, further supporting the idea of a shared genetic basis across various psychiatric conditions (Marees et al., 2021).
These findings from GWAS not only highlight the importance of common genetic variants but also pave the way for the development of polygenic risk scores (PRS), which aggregate the effects of multiple genetic variants to estimate an individual’s genetic risk for developing a particular disorder. Although still in its early stages, PRS holds promise for future clinical applications, potentially aiding in the early identification of individuals at higher risk for psychiatric disorders (Brainstorm Consortium et al., 2018).
Epigenetics: Bridging the Gap Between Genes and Environment
Epigenetics serves as the interface between our genetic code and the environment, mediating how external factors can influence gene expression and, consequently, behavior. Unlike genetic mutations, which alter the DNA sequence itself, epigenetic changes modify the structure or accessibility of DNA, thereby influencing how genes are expressed without altering the genetic code. This regulatory system involves several key mechanisms: DNA methylation, histone modification, and the action of non-coding RNAs (ncRNAs).
DNA methylation is perhaps the most extensively studied epigenetic mechanism. It involves the addition of a methyl group to the cytosine base in CpG dinucleotides, typically leading to gene repression. Methylation patterns are established during development and can be altered by environmental factors such as diet, stress, and exposure to toxins. These modifications are heritable during cell division, which means that the effects of environmental exposures can persist across multiple cell generations, potentially even being passed on to offspring (Johnston et al., 2024). In the brain, aberrant DNA methylation has been linked to various psychiatric disorders, including schizophrenia and major depressive disorder, by disrupting the normal expression of genes involved in synaptic function and neuroplasticity (Moore et al., 2013).
Histones are proteins around which DNA is wrapped, forming the basic unit of chromatin known as the nucleosome. Post-translational modifications of histone tails, such as methylation, acetylation, phosphorylation, and ubiquitination, play crucial roles in regulating gene expression. For instance, histone acetylation, typically associated with transcriptional activation, loosens the chromatin structure, making DNA more accessible to transcription factors. Conversely, histone methylation can either activate or repress gene expression, depending on the specific residues that are modified (Kreibich et al., 2023). For example, trimethylation of histone H3 at lysine 4 (H3K4me3) is generally associated with active transcription, whereas trimethylation at lysine 27 (H3K27me3) is linked to gene silencing. These modifications are dynamic and can be influenced by environmental factors, thereby providing a mechanism for the environment to exert long-term effects on gene expression and behavior (Moore et al., 2013; Emileva Krasteva, 2024).
NcRNAs, including microRNAs (miRNAs) and long non-coding RNAs (lncRNAs), have emerged as important regulators of gene expression, often through interactions with other epigenetic mechanisms. MiRNAs can silence gene expression by binding to messenger RNA (mRNA) and preventing its translation, while lncRNAs can modulate gene expression by recruiting chromatin-modifying complexes to specific genomic loci (Eriksson et al., 2014). For instance, the miRNA miR-137 has been implicated in schizophrenia, where it is thought to regulate the expression of genes involved in synaptic plasticity and neurodevelopment. Additionally, lncRNAs can serve as scaffolds for histone modification complexes, thereby influencing chromatin structure and gene expression on a broader scale.
One of the most compelling aspects of epigenetics is its role in mediating the effects of environmental exposures on behavior. Factors such as stress, diet, and exposure to toxins can lead to epigenetic modifications that alter gene expression patterns in the brain, potentially leading to changes in behavior and increasing the risk of psychiatric disorders. For example, early life stress has been shown to induce lasting changes in DNA methylation and histone modification patterns in genes related to stress response, such as the glucocorticoid receptor gene, which is crucial for regulating the hypothalamic-pituitary-adrenal (HPA) axis (Emileva Krasteva, 2024). These epigenetic changes can result in altered stress reactivity and increased vulnerability to mood disorders later in life.
Epigenetics in Specific Behavioral Disorders
Epigenetic research has increasingly illuminated the complex interplay between genetic predispositions and environmental factors in the etiology of various psychiatric disorders. In the context of mood disorders, such as depression and anxiety, epigenetic mechanisms like DNA methylation, histone modification, and non-coding RNAs play critical roles in regulating gene expression without altering the underlying DNA sequence. For instance, studies have shown that chronic stress, a known risk factor for depression and anxiety, can lead to persistent changes in DNA methylation patterns. These modifications influence the expression of genes involved in the stress response, potentially leading to the development and maintenance of mood disorders (Azargoonjahromi, 2023).
One of the most studied epigenetic changes in mood disorders involves the regulation of the glucocorticoid receptor gene (NR3C1). This gene is pivotal in the body’s response to stress, and its expression is often found to be altered in individuals with depression and anxiety, particularly those with a history of early-life stress. For example, hypermethylation of the NR3C1 promoter has been associated with reduced gene expression and an exaggerated stress response, a hallmark of depression (Jones et al., 2021). Furthermore, epigenetic modifications can also affect the efficacy of antidepressant treatments. Histone modifications, such as the acetylation of histones H3 and H4, have been implicated in the therapeutic response to antidepressants, indicating that epigenetic mechanisms might also contribute to treatment resistance in some patients (Peedicayil, 2020).
Schizophrenia, another major psychiatric disorder, is also increasingly understood through the lens of epigenetics. Research has identified several epigenetic alterations associated with schizophrenia, including changes in DNA methylation and histone modification that affect genes involved in neural development and synaptic function. For instance, hypermethylation of the RELN gene, which encodes the protein reelin, is frequently observed in schizophrenia patients. Reelin is crucial for neuronal migration and synaptic plasticity, and its reduced expression due to epigenetic silencing is thought to contribute to the neurodevelopmental aspects of schizophrenia (Kular & Kular, 2018).
Moreover, genome-wide association studies (GWAS) have revealed that many of the genetic loci associated with schizophrenia are located in regions of the genome that are subject to epigenetic regulation. These findings suggest that epigenetic mechanisms could mediate the effects of genetic risk factors on gene expression, thereby influencing the onset and progression of schizophrenia (Ovenden et al., 2018). In addition to these molecular findings, studies have also highlighted the role of environmental factors, such as prenatal stress and early-life adversity, in shaping the epigenetic landscape in ways that increase susceptibility to schizophrenia later in life (Rutten & Mill, 2009).
The Role of Early Life Experiences in Epigenetic Modifications
Early life experiences, particularly during prenatal and early childhood stages, are crucial in shaping long-term behavioral outcomes through epigenetic modifications. Epigenetics refers to heritable changes in gene expression that do not involve alterations in the DNA sequence itself, and these changes can be profoundly influenced by environmental factors such as maternal stress and nutrition during critical developmental periods.
Prenatal exposure to maternal stress, for example, has been extensively studied for its impact on the epigenome of the developing fetus. Stress-related hormonal changes in the mother, particularly elevated levels of cortisol, can lead to modifications in DNA methylation patterns in the fetus. These epigenetic changes can affect genes related to stress response, such as the glucocorticoid receptor gene (NR3C1), which plays a pivotal role in regulating the hypothalamic-pituitary-adrenal (HPA) axis. Dysregulation of this axis is commonly associated with mood and anxiety disorders later in life. Notably, studies have shown that children exposed to high levels of maternal stress in utero exhibit altered DNA methylation in these key genes, which is associated with increased vulnerability to behavioral disorders such as depression and anxiety during adolescence and adulthood (Conradt et al., 2013; Devlin et al., 2010).
Nutrition during early life also significantly influences epigenetic marks, which in turn can have lasting effects on brain development and function. For instance, deficiencies in critical nutrients like iron, folate, and choline during pregnancy can lead to altered methylation of genes involved in neurodevelopmental processes. Iron deficiency, in particular, has been linked to impaired hippocampal development, which is crucial for memory and learning. These epigenetic alterations can persist beyond the period of deficiency, potentially contributing to cognitive deficits and increased risk of neurodevelopmental disorders such as autism and schizophrenia later in life (Haggarty, 2012; Siddappa et al., 2004).
Moreover, the timing of these environmental exposures is critical. The brain is especially vulnerable to epigenetic influences during periods of rapid growth and development, such as in the prenatal stage and early childhood. Nutritional imbalances or maternal stress during these sensitive windows can result in epigenetic modifications that alter the expression of genes responsible for synaptic plasticity, neurogenesis, and neurotransmitter systems. These changes can affect brain architecture and function, leading to long-term behavioral and psychological outcomes (Hensch, 2005).
Transgenerational Epigenetic Inheritance and Behavioral Disorders
Transgenerational epigenetic inheritance refers to the phenomenon where environmental factors, such as stress or trauma, lead to epigenetic modifications that are passed down through generations, potentially influencing the behavior and mental health of offspring. These epigenetic changes, including DNA methylation and histone modifications, can be transmitted via germ cells (sperm and eggs) without altering the underlying DNA sequence. For example, research has shown that stress-induced epigenetic changes in one generation can result in altered stress responses and increased susceptibility to psychiatric disorders, such as anxiety and depression, in subsequent generations (Crews et al., 2012; McEwen et al., 2012).
One of the key mechanisms underlying transgenerational epigenetic inheritance involves the regulation of genes associated with stress responses and neural development. For instance, epigenetic modifications to the glucocorticoid receptor gene (NR3C1) in response to parental trauma can lead to altered HPA axis activity in offspring, which is linked to increased vulnerability to stress-related disorders. Moreover, studies in rodents have demonstrated that exposure to environmental toxins or stressors can cause epigenetic alterations that are inherited by multiple generations, leading to behavioral changes and increased risk of psychiatric disorders (Cao-Lei et al., 2021).
The implications of transgenerational epigenetic inheritance are profound, particularly in understanding the heritability of psychiatric disorders. Traditional genetic models often fail to fully explain the familial patterns observed in disorders like schizophrenia and bipolar disorder. However, the recognition that epigenetic marks can be inherited across generations provides a more comprehensive understanding of how environmental factors and genetic predispositions interact to influence mental health. This paradigm shift highlights the need for psychiatric research to consider both genetic and epigenetic factors when studying the etiology and transmission of behavioral disorders (Bohacek & Mansuy, 2015).
Additionally, this concept raises important questions about the potential for reversing or mitigating these inherited epigenetic changes. If epigenetic marks can be modified by environmental factors, there may be therapeutic potential in interventions that target these mechanisms to prevent the transmission of vulnerability to psychiatric disorders across generations (Korolenko et al., 2023).
Epigenetics and Treatment: Implications for Psychiatry
Epigenetic research has significant potential to transform the landscape of psychiatric treatment by paving the way for personalized medicine. Personalized medicine in psychiatry involves tailoring treatments based on an individual’s genetic and epigenetic profile, allowing for more targeted and effective interventions. The understanding of how epigenetic mechanisms, such as DNA methylation and histone modification, influence gene expression has provided insights into the pathophysiology of various psychiatric disorders, including depression, schizophrenia, and anxiety disorders. These insights can be leveraged to develop novel therapeutic strategies that modify epigenetic marks, thus influencing the underlying genetic predispositions that contribute to these disorders (Grezenko et al., 2023).
One promising area of research is the use of histone deacetylase (HDAC) inhibitors as potential therapeutic agents. HDAC inhibitors work by altering the acetylation status of histones, thereby affecting chromatin structure and gene expression. These inhibitors have shown promise in preclinical studies for treating conditions such as depression and schizophrenia. For instance, the modulation of histone acetylation has been linked to the reversal of stress-induced behavioral changes in animal models, suggesting a potential avenue for therapeutic intervention in humans (Abel & Zukin, 2008). Moreover, ongoing clinical trials are exploring the efficacy of HDAC inhibitors in combination with traditional pharmacotherapies, aiming to enhance the overall therapeutic outcomes for patients with psychiatric disorders (Sahafnejad et al., 2023).
However, the integration of epigenetic therapies into clinical practice is not without challenges. One of the primary concerns is the dynamic nature of the epigenome, which can change in response to environmental factors, age, and even the course of the disease itself. This variability makes it difficult to develop stable and predictive biomarkers for clinical use. Additionally, there are ethical considerations related to epigenetic treatments, particularly concerning the potential for unintended consequences. For example, while modifying epigenetic marks could alleviate symptoms of a psychiatric disorder, there is also the risk of off-target effects or long-term changes that might affect other aspects of an individual’s health (Santaló & Berdasco, 2022).
Ethical considerations also extend to issues of patient consent and autonomy. As epigenetic data becomes increasingly integrated into personalized treatment plans, healthcare providers must navigate the complexities of communicating these risks to patients. The International Human Epigenome Consortium (IHEC) emphasizes the need for clear guidelines on the communication of epigenetic information to patients, ensuring that they are fully informed about the potential risks and benefits of such treatments. Moreover, there is a need for ongoing ethical oversight as the field of epigenetics in psychiatry continues to evolve, particularly in relation to the potential for epigenetic interventions to influence not just the individual but potentially future generations (Yuan et al., 2023).
Future Directions and Challenges in Epigenetic Research in Psychiatry
Emerging technologies and ethical considerations are central to the future directions and challenges in epigenetic research in psychiatry. One of the most promising advances is single-cell epigenomics, which allows for the analysis of epigenetic modifications at the resolution of individual cells. This technology offers significant advantages over traditional bulk sequencing methods, which often obscure the complexity of epigenetic landscapes by averaging signals across heterogeneous cell populations. Single-cell approaches, such as single-cell bisulfite sequencing (scBS-seq) and single-cell Hi-C, have been instrumental in uncovering the variability of epigenetic marks, such as DNA methylation and chromatin conformation, among individual cells. These tools are poised to revolutionize our understanding of how epigenetic modifications contribute to psychiatric disorders by providing a more detailed and nuanced view of gene regulation at the cellular level (Clark et al., 2016).
However, integrating these cutting-edge technologies into clinical practice remains a significant challenge. The vast amount of data generated by single-cell epigenomics and other high-resolution methods necessitates sophisticated bioinformatics tools for analysis and interpretation. Moreover, the complexity of the epigenome, which is highly dynamic and varies across different tissues, developmental stages, and environmental contexts, complicates the translation of these findings into actionable clinical insights (Deans & Maggert, 2015; Silveira et al., 2007).
Ethical considerations also play a critical role in the application of epigenetic research in psychiatry. The use of epigenetic biomarkers for disease prediction and personalized treatment raises concerns about data privacy, the potential for genetic discrimination, and the psychological impact on patients. For example, communicating the results of epigenetic tests to patients requires careful consideration of how this information is presented, given the complexities and uncertainties associated with epigenetic data. There is also a need for guidelines and policies to ensure that epigenetic information is used ethically and that patients’ rights and autonomy are respected (Sánchez-Mut et al., 2014; Hegarty et al., 2014).
As the field continues to evolve, addressing these challenges will be crucial for harnessing the full potential of epigenetic research in psychiatry. Advances in single-cell technologies, combined with a robust ethical framework, could pave the way for more precise and personalized approaches to treating psychiatric disorders, ultimately improving patient outcomes and advancing our understanding of the intricate interplay between genetics, epigenetics, and behavior (Feil & Fraga, 2012; Atun et al., 2015).
Conclusion
In conclusion, this literature review has explored the intricate relationship between genetics, epigenetics, and behavioral disorders, highlighting the growing recognition of epigenetic mechanisms as crucial mediators between genes and the environment. The evidence presented underscores the significance of early life experiences, such as prenatal stress and childhood adversity, in shaping long-term behavioral outcomes through epigenetic modifications. The review also emphasized the potential for epigenetic research to revolutionize psychiatric treatment by informing personalized medicine and developing novel therapeutic approaches.
While this review focused on the biological and epigenetic underpinnings of behavioral disorders, it is important to recognize that successful psychiatric care often requires a holistic approach. Integrating psychotherapy with insights from genetic and epigenetic research can significantly enhance personalized treatment strategies. Psychotherapy, tailored to the unique biological and psychological profiles of individuals, has the potential to complement personalized medication, addressing both the underlying biological mechanisms and the psychological needs of patients.
However, the challenges of integrating these insights into clinical practice, alongside ethical considerations, remain substantial. As emerging technologies like single-cell epigenomics continue to advance, the potential for more precise and individualized interventions in psychiatry becomes increasingly feasible. This combined approach—merging biological, epigenetic, and psychotherapeutic strategies—promises to enhance our understanding and treatment of complex behavioral disorders, paving the way for a truly personalized and holistic paradigm in psychiatric care.
References
Abel, T. & Zukin, R. S. (2008). Epigenetic targets of HDAC inhibition in neurodegenerative and psychiatric disorders. Current opinion in pharmacology, 8(1), 57–64. https://doi.org/10.1016/j.coph.2007.12.002
Andreassen, O. A. et al. (2023). New insights from the last decade of research in psychiatric genetics: discoveries, challenges and clinical implications. World psychiatry: official journal of WPA, 22(1), 4–24. https://doi.org/10.1002/wps.21034
Atun, R. et al. (2015). Expanding global access to radiotherapy. The Lancet. Oncology, 16(10), 1153–1186. https://doi.org/10.1016/S1470-2045(15)00222-3
Azargoonjahromi, A. (2023). The role of epigenetics in anxiety disorders. Molecular biology reports, 50(11), 9625–9636. https://doi.org/10.1007/s11033-023-08787-6
Bohacek, J. & Mansuy, I. M. (2015). Molecular insights into transgenerational non-genetic inheritance of acquired behaviours. Nature reviews. Genetics, 16(11), 641–652. https://doi.org/10.1038/nrg3964
Brainstorm Consortium et al. (2018). Analysis of shared heritability in common disorders of the brain. Science (New York, N. Y.), 360(6395), eaap8757. https://doi.org/10.1126/science.aap8757
Cao-Lei, L. et al. (2021). Epigenetic Modifications Associated with Maternal Anxiety during Pregnancy and Children’s Behavioral Measures. Cells, 10(9), 2421. https://doi.org/10.3390/cells10092421
Clark, S. J. et al. (2016). Single-cell epigenomics: powerful new methods for understanding gene regulation and cell identity. Genome biology, 17, 72. https://doi.org/10.1186/s13059-016-0944-x
Conradt, E. et al. (2013). The roles of DNA methylation of NR3C1 and 11β-HSD2 and exposure to maternal mood disorder in utero on newborn neurobehavior. Epigenetics, 8(12), 1321–1329. https://doi.org/10.4161/epi.26634
Crews, D. et al. (2012). Epigenetic transgenerational inheritance of altered stress responses. Proceedings of the National Academy of Sciences of the United States of America, 109(23), 9143–9148. https://doi.org/10.1073/pnas.1118514109
Deans, C. & Maggert, K. A. (2015). What do you mean, «epigenetic»? Genetics, 199(4), 887–896. https://doi.org/10.1534/genetics.114.173492
Devlin, A. M. et al. (2010). Prenatal exposure to maternal depressed mood and the MTHFR C677T variant affect SLC6A4 methylation in infants at birth. PloS one, 5(8), e12201. https://doi.org/10.1371/journal.pone.0012201
Emileva Krasteva, M. (2024). Environmental Stress, Epigenetic Modifications, Adaptation, and Disease: A Fine Interplay. IntechOpen. https://doi.org/10.5772/intechopen.1004285
Eriksson, M. et al. (2014). Early life stress and later health outcomes. Findings from the Helsinki Birth Cohort Study. American journal of human biology: the official journal of the Human Biology Council, 26(2), 111–116. https://doi.org/10.1002/ajhb.22502
Feil, R. & Fraga, M. F. (2012). Epigenetics and the environment: emerging patterns and implications. Nature reviews. Genetics, 13(2), 97–109. https://doi.org/10.1038/nrg3142
Grezenko, H. et al. (2023). Epigenetics in Neurological and Psychiatric Disorders: A Comprehensive Review of Current Understanding and Future Perspectives. Cureus, 15(8), e43960. https://doi.org/10.7759/cureus.43960
Guo, Y. et al. (2024). Associations between ADHD and risk of six psychiatric disorders: a Mendelian randomization study. BMC psychiatry, 24(1), 99. https://doi.org/10.1186/s12888-024-05548-y.
Haggarty, P. (2012). Nutrition and the epigenome. Progress in molecular biology and translational science, 108, 427–446. https://doi.org/10.1016/B978-0-12-398397-8.00016-2
Hegarty, S. V. et al. (2014). Neurotrophic factors: from neurodevelopmental regulators to novel therapies for Parkinson’s disease. Neural regeneration research, 9(19), 1708–1711. https://doi.org/10.4103/1673-5374.143410
Hensch, T. K. (2005). Critical period plasticity in local cortical circuits. Nature reviews. Neuroscience, 6(11), 877–888. https://doi.org/10.1038/nrn1787
Johnston, R. A. et al. (2024). DNA methylation-environment interactions in the human genome. eLife, 12, RP89371. https://doi.org/10.7554/eLife.89371
Jones, D. E. et al. (2021). Mental health epigenetics: A primer with implications for counselors. The Professional Counselor, 11(1), 102–121. https://doi.org/10.15241/dej.11.1.102
Korolenko, A. A. et al. (2023). Epigenetic Inheritance and Transgenerational Environmental Justice. The Yale journal of biology and medicine, 96(2), 241–250. https://doi.org/10.59249/FKWS5176
Kreibich, E. et al. (2023). Single-molecule footprinting identifies context-dependent regulation of enhancers by DNA methylation. Molecular cell, 83(5), 787–802.e9. https://doi.org/10.1016/j.molcel.2023.01.017
Kular, L. & Kular, S. (2018). Epigenetics applied to psychiatry: Clinical opportunities and future challenges. Psychiatry and clinical neurosciences, 72(4), 195–211. https://doi.org/10.1111/pcn.12634
Marees, A. T. et al. (2021). Genetic correlates of socio-economic status influence the pattern of shared heritability across mental health traits. Nature human behaviour, 5(8), 1065–1073. https://doi.org/10.1038/s41562-021-01053-4
McEwen, B. S. et al. (2012). Stress and anxiety: structural plasticity and epigenetic regulation as a consequence of stress. Neuropharmacology, 62(1), 3–12. https://doi.org/10.1016/j.neuropharm.2011.07.014
Moore, L. D. et al. (2013). DNA methylation and its basic function. Neuropsychopharmacology, 38(1), 23–38. https://doi.org/10.1038/npp.2012.112
Ovenden, E. S. et al. (2018). DNA methylation and antipsychotic treatment mechanisms in schizophrenia: Progress and future directions. Progress in neuro-psychopharmacology & biological psychiatry, 81, 38–49. https://doi.org/10.1016/j.pnpbp.2017.10.004
Peedicayil, J. (2020). The Potential Role of Epigenetic Drugs in the Treatment of Anxiety Disorders. Neuropsychiatric disease and treatment, 16, 597–606. https://doi.org/10.2147/NDT.S242040
Pettersson, E. et al. (2016). Common psychiatric disorders share the same genetic origin: a multivariate sibling study of the Swedish population. Molecular psychiatry, 21(5), 717–721. https://doi.org/10.1038/mp.2015.116
Pina-Camacho, L. et al. (2018). Autism spectrum disorder and schizophrenia: boundaries and uncertainties. BJPsych Advances, 22(5), 316–324. https://doi.org/10.1192/apt.bp.115.014720
Ribasés, M. et al. (2023). Genetic architecture of ADHD and overlap with other psychiatric disorders and cognition-related phenotypes. Neuroscience and biobehavioral reviews, 153, 105313. https://doi.org/10.1016/j.neubiorev.2023.105313
Rutten, B. P. & Mill, J. (2009). Epigenetic mediation of environmental influences in major psychotic disorders. Schizophrenia bulletin, 35(6), 1045–1056. https://doi.org/10.1093/schbul/sbp104
Sanchez-Mut, J. V. et al. (2014). Promoter hypermethylation of the phosphatase DUSP22 mediates PKA-dependent TAU phosphorylation and CREB activation in Alzheimer’s disease. Hippocampus, 24(4), 363–368. https://doi.org/10.1002/hipo.22245
Santaló, J. & Berdasco, M. (2022). Ethical implications of epigenetics in the era of personalized medicine. Clinical epigenetics, 14(1), 44. https://doi.org/10.1186/s13148-022-01263-1
Sahafnejad, Z. et al. (2023). An Update of Epigenetic Drugs for the Treatment of Cancers and Brain Diseases: A Comprehensive Review. Genes, 14(4), 873. https://doi.org/10.3390/genes14040873
Siddappa, A. M. et al. (2004). Iron deficiency alters auditory recognition memory in newborn infants of diabetic mothers. Pediatric research, 55(6), 1034–1041. https://doi.org/10.1203/01.pdr.0000127021.38207.62
Silveira, P. P. et al. (2007). Developmental origins of health and disease (DOHaD). Jornal de pediatria, 83(6), 494–504. https://doi.org/10.2223/JPED.1728
Wang, D. G. et al. (1998). Large-scale identification, mapping, and genotyping of single-nucleotide polymorphisms in the human genome. Science (New York, N. Y.), 280(5366), 1077–1082. https://doi.org/10.1126/science.280.5366.1077
Yuan, M. et al. (2023). Epigenetic regulation in major depression and other stress-related disorders: molecular mechanisms, clinical relevance and therapeutic potential. Sig Transduct Target Ther, 8, 309. https://doi.org/10.1038/s41392-023-01519-z
L’architettura genetica dei disturbi del comportamento
Esplorare il ruolo dell’epigenetica in psichiatria
Riassunto: I disturbi comportamentali come la depressione, la schizofrenia, l’ADHD e i disturbi dello spettro autistico rappresentano una sfida significativa per la psichiatria a causa della loro natura complessa e multifattoriale. Mentre i modelli tradizionali si sono concentrati sulle predisposizioni genetiche, le ricerche emergenti evidenziano il ruolo critico dell’epigenetica nel modulare l’espressione genica in risposta ai fattori ambientali. Questa rassegna della letteratura esplora l’architettura genetica dei disturbi comportamentali, sottolineando l’influenza dei meccanismi epigenetici come la metilazione del DNA, la modificazione degli istoni e gli RNA non codificanti. Si analizza il modo in cui questi processi contribuiscono all’insorgenza, alla progressione e alla trasmissione transgenerazionale delle patologie psichiatriche. Inoltre, discutiamo le implicazioni di questi risultati per le strategie di trattamento personalizzate e le future direzioni di ricerca, sottolineando il potenziale delle terapie epigenetiche nel rimodellare la pratica psichiatrica. Questa rassegna mira a colmare il divario tra genetica e ambiente, offrendo una comprensione completa di come l’epigenetica possa informare il futuro delle cure psichiatriche.
Parole chiave: disturbi comportamentali, epigenetica, architettura genetica, psichiatria, medicina personalizzata
Biographical Note
Kirolos Eskandar, Physician, is affiliated with the Diakonie Klinik Mosbach, Germany. He is an independent researcher with a strong academic foundation, holding a Bachelor of Science, a Master of Arts, and a Bachelor of Medicine and Surgery. His clinical experience spans various specialties, including neuropsychiatry and internal medicine, with a focus on diagnosing and treating mental health conditions. He is an author of many articles in the areas such as Medical advancements, Oncology, Psychiatry and serves on the editorial board of the World Journal of Health and Medicine. ORCID ID: 0000-0003-0085-3284
Contact